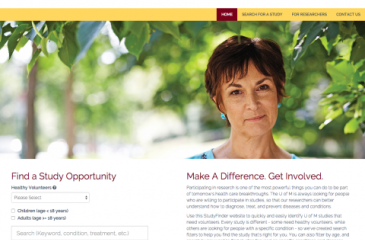
The University of Minnesota’s health research website, StudyFinder, was recently adopted, customized, and launched by Penn State University, which makes it the first institution outside the University to adopt the tool. In addition, Virginia Commonwealth University (VCU) launched its StudyFinder website today for use in that community.
The site, built by the University’s Clinical and Translational Science Institute’s Recruitment Center and the Academic Health Center’s Information Systems department, was developed in 2013 as a way for the public to explore and connect with University of Minnesota research studies.
StudyFinder displays a searchable listing of University of Minnesota studies, which are extracted from ClinicalTrials.gov, an international registry of clinical trials involving human participants from 50 states and 192 countries, which is run by the United States National Library of Medicine (NLM) at the National Institutes of Health (NIH).
“StudyFinder extracts the data, which is updated daily, from ClinicalTrials.gov and displays it in a way that makes it easy for the general public to understand what a particular study is focused on, and find a research opportunity that’s right for them,” said Melissa Mueller, MPH, CTSI’s Recruitment Service Manager and the tool’s co-creater. “StudyFinder's simple interface makes it easier for Minnesotans to participate in research and help develop treatments that may one day benefit a friend, a family member, or someone around the world.”
StudyFinder also has the technical capabilities to pull in data from other sources, such as OnCore clinical trials management system.
“Studies are looking for participants of all ages and health conditions, even healthy volunteers who don’t have the condition being studied. With more than 360 studies currently listed on the University of Minnesota’s StudyFinder website, there’s a research opportunity for every Minnesotan,” Mueller said.
Helping increase research discoveries nationwide
The University’s CTSI, through its National Center for Advancing Translational Sciences grant, joined a consortium of Clinical and Translational Science Awards (CTSA) sites, which includes more than 50 institutions across the nation that work together to improve the translational research process to provide more treatments to more patients more quickly.
One way in which this goal is being met is through the the University’s StudyFinder tool.
“The site was built in open-source code so other CTSA sites from across the country can easily adopt it for their institutions and communities,” said Mueller. “Shared infrastructure that can be easily exported to other CTSA sites ultimately means less time spent developing the infrastructure and more time spent removing other barriers that prevent health breakthroughs from benefitting the public.”
Other CTSA institutions are also engaging in discussions with CTSI about adopting StudyFinder.