News

StudyFinder tool continues national expansion enabling public to explore studies at academic research institutions
University of Texas Southwestern has become the third research institution to adopt StudyFinder, a CTSI-created tool that gives the public an easier way to find university research opportunities currently enrolling and connect with study…
CTSI enters final stage of TASCS-OnCore transition
This month, CTSI began the final phase of an ongoing, collaborative effort to shift University of Minnesota clinical research studies from the Time and

CTSI awards more than $9,000 to seven researchers to present at major conferences
The Clinical and Translational Science Institute (CTSI) recognized researchers for outstanding posters and presentations at the annual CTSI Poster Session and Reception held September 20.More than 30 research faculty and community judges…
Electronic case report forms now available through OnCore
More OnCore clinical trial management system users can now capture visit-level data, thanks to the availability of electronic case report forms (eCRFs) across the Academic Health Center. OnCore eCRFs were previously only available to the…
OnCore replaces TASCS across the AHC for new studies
Academic Health Center (AHC) researchers will no longer enter new studies into the Time and Study Collection System (TASCS), thanks to the transition to the OnCore clinical trial management system.
After Oct. 1, TASCS -- previously used…
CTSI sharpens its invoicing infrastructure to ensure timely, quality billing of research services
Over the last few months, CTSI has reviewed and refined its invoice functionality within the Clinical and Translational (CTR) Portal for research services performed by a university researcher. During this review, deficits were found,…
CTSI pilots new tool for engaging communities to inform your research
CTSI’s Office of Community Engagement to Advance Research and Community Health (CEARCH) recently successfully piloted the Community Engagement Studio model aimed at better engaging the community to help inform research conducted at the…
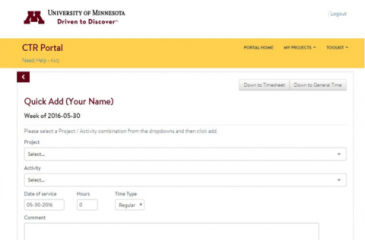
Time Sheet module now offers more dynamic and user-friendly experience
CTSI has enhanced the Clinical Translational Research Portal's (CTR Portal) Time Sheet module to create a better experience for the University of Minnesota research teams who use it.
CTSI team members and select service units and…
Research services enhancements help increase administrative, invoicing efficiencies
Beginning next week, the DeptID for the department administering the research project will be required for all new requests for services submitted in CTSI’s Clinical and Translational Research (CTR) Portal and project set-up requests made…
